AtomLens
AtomLens helps you read and understand molecule and reaction files on macOS. Open common chemistry formats, inspect clean 2D structures and reaction schemes, compute identifiers and key properties, split multi-entry files when needed, and find results quickly with Spotlight and Quick Look.
What it does
- Open chemistry formats such as MOL, SDF, SMILES, Reaction SMILES, InChI, MOL2, PDB, XYZ, CML, RXN, and RDF.
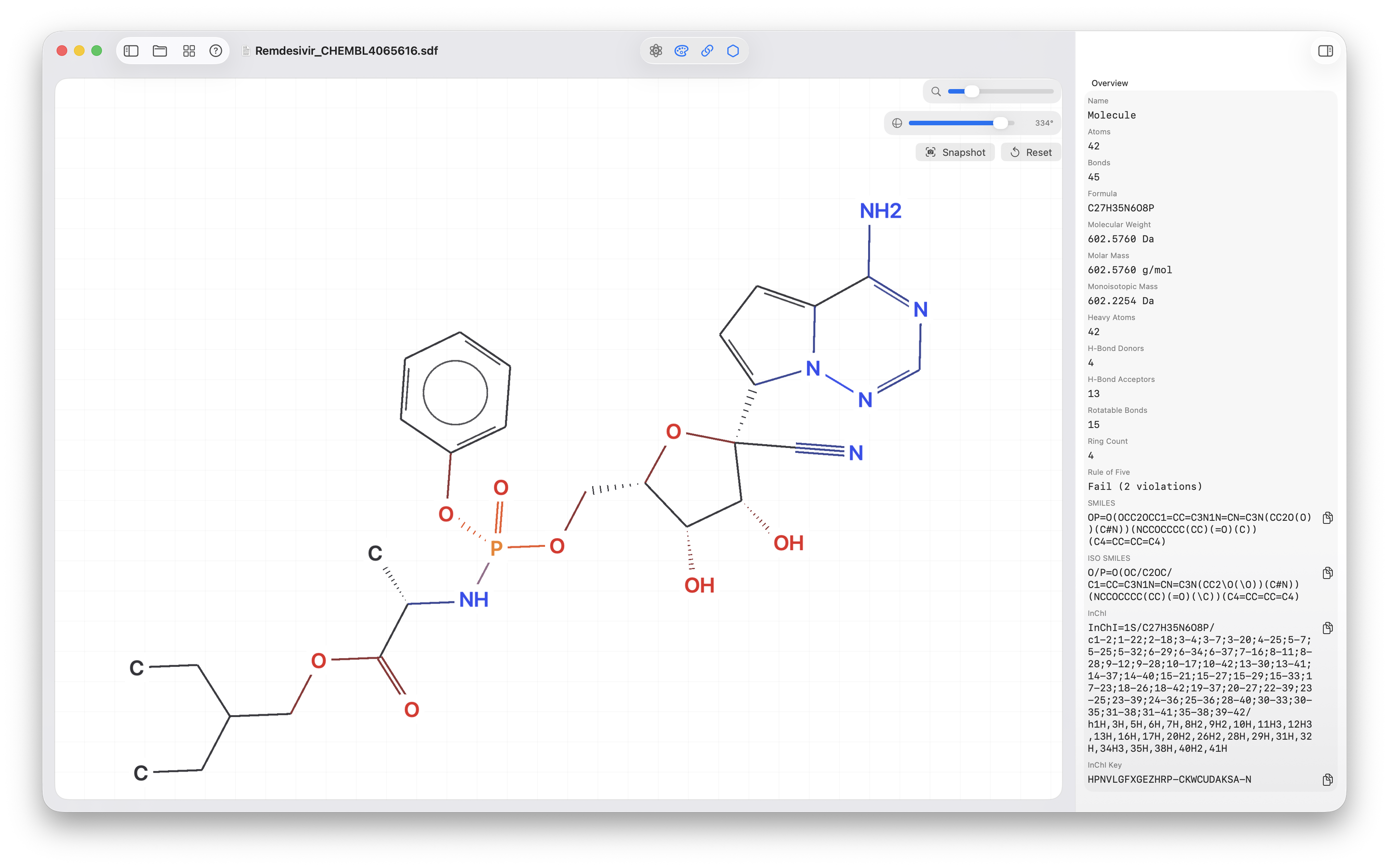
- Render clean 2D molecule views and dedicated reaction views in a native macOS interface.
- Generate identifiers: SMILES, isomeric SMILES, InChI, and InChI Key.
- Calculate core properties like formula, molecular weight, monoisotopic mass, LogP, and Rule-of-Five status.
- Split multi-structure SDF and multi-reaction SMILES files into individual outputs for downstream work.
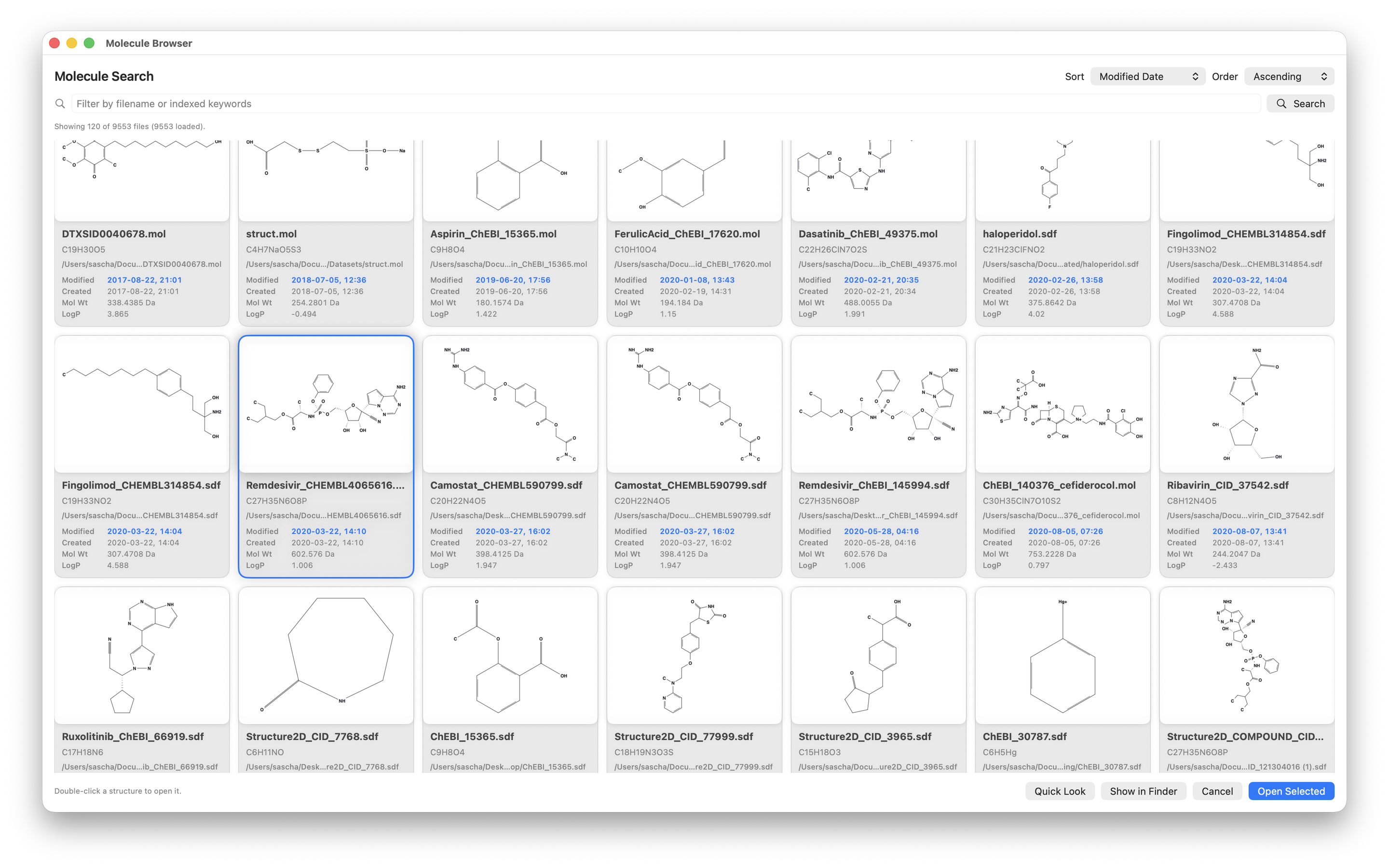
- Search molecule and reaction files locally through Spotlight metadata and preview them with Quick Look.
Tip: The Help page includes practical search queries and full format tables.
Designed for local workflows
AtomLens 1.1 expands day-to-day lab and research workflows from single structures into reactions and extraction jobs. You can inspect files, review participant context, export in common formats, and keep all data on your Mac.
No cloud upload required for standard workflows.
What's new in Version 1.1
AtomLens 1.1 focuses on reaction workflows, extraction tools, and overall reliability across native macOS use.
- Added full Reaction SMILES (
.rsmi) support across open, preview, and Spotlight indexing flows. - Added a dedicated reaction document view with reactants, agents, and products shown in context.
- Added Extract tools to split multi-structure SDF files, split multi-reaction SMILES into
.rsmi, and convert them into.rxnfiles. - Improved parse and export progress windows, clearer error reporting, and smoother window restoration and file-open behavior.
- Updated Quick Look, Spotlight integration, the app icon, and the published Help content.
Try sample structures
Use this starter set of molecule and reaction files to test parsing, rendering, extraction, and Spotlight/Quick Look workflows in AtomLens.
Download example structures (ZIP)
Stable link for future site updates: /assets/downloads/atomlens-example-structures.zip
Screenshots
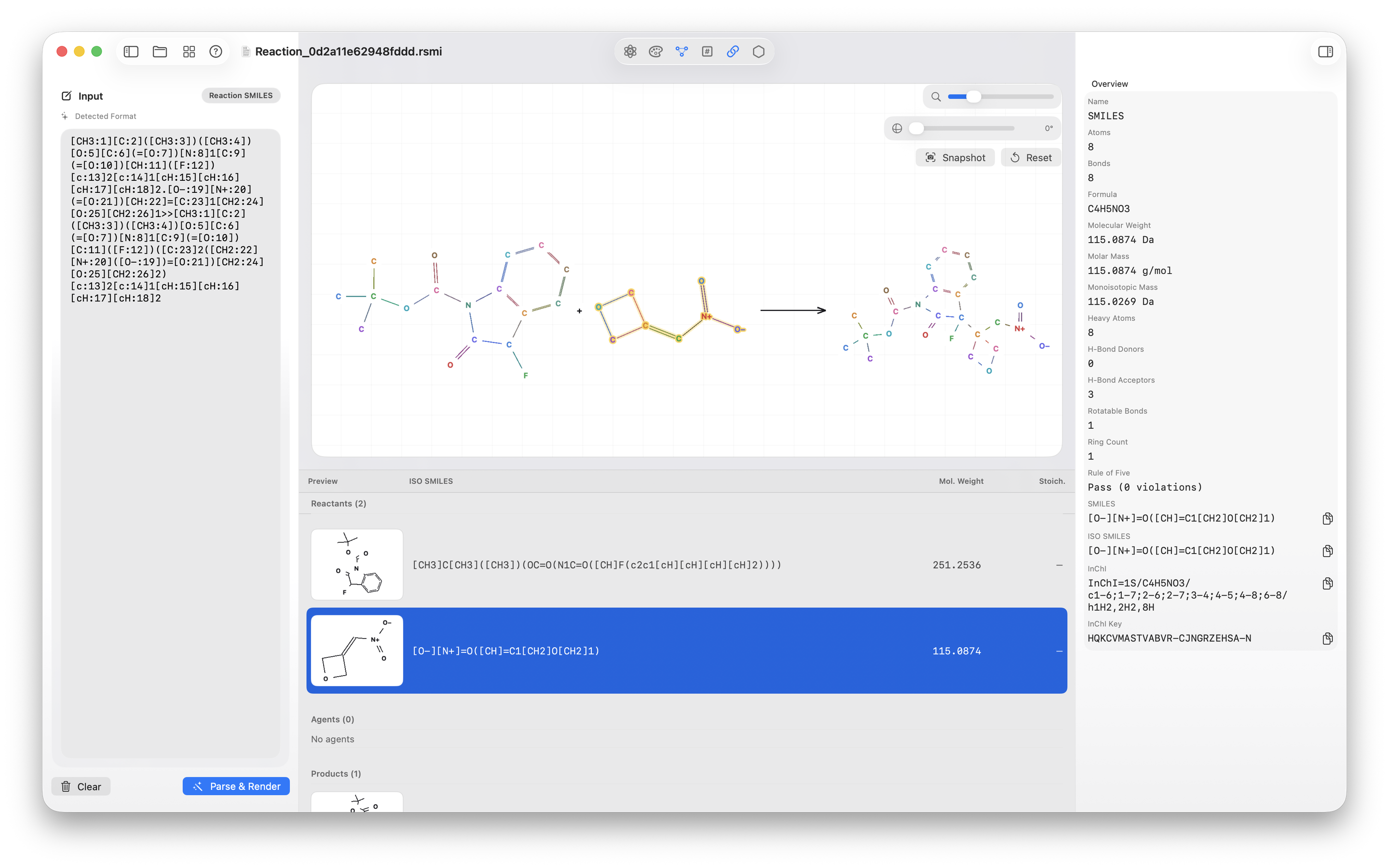
.rsmi, review reactants, agents, and products, and inspect participant details.The spark: why this app exists
If you work with molecules, you know the moment: someone sends "the compound" as an SDF attachment, your downloads folder becomes a museum, and a week later you remember it had an InChI Key — but not the filename. Traditional cheminformatics tools are powerful, but the operating system itself still treats molecule files like anonymous text.
AtomLens started as a simple wish: "Finder should be able to preview and search molecules like it previews and searches PDFs." From that, a surprisingly ambitious requirements list fell out:
- Open common chemistry formats: SDF, MOL, SMILES, InChI, and more.
- Render chemist-friendly 2D depictions: aromatic rings read as aromatic, chains are readable, stereo bonds are unambiguous.
- Compute identifiers and properties: SMILES, ISO SMILES, InChI, InChI Key, formula, molecular weight, LogP, Rule of Five.
- Index those values into Spotlight and show them in Finder via Quick Look previews and thumbnails.
- Remain App Store compliant: sandbox-safe, predictable permissions, and no external converter dependencies.
Spotlight, Quick Look, and the macOS "superpowers"
This is the part that makes AtomLens feel "native" in a way most scientific tools do not: structures become searchable and previewable without opening the app. Spotlight indexing adds metadata to molecule and reaction files (SMILES, InChI, InChI Key, formula, weight, LogP, and reaction flags), and Quick Look turns Spacebar into an instant chemistry preview.
We also built a Molecule Browser that queries Spotlight directly. It is essentially a local chemistry-library UI without a separate database - just the macOS index you already have.
Chemistry engine
AtomLens is powered by CDKSwiftNativePort, a Swift-native chemistry toolkit derived from the original Chemistry Development Kit (CDK). It drives molecule and reaction parsing, depiction, identifiers, and property calculations locally on macOS.
Get help
Full documentation is available here: AtomLens Help.
For feedback and bug reports, email support@losko.de.
